Process Analytical Technology
Process Analytical Technology
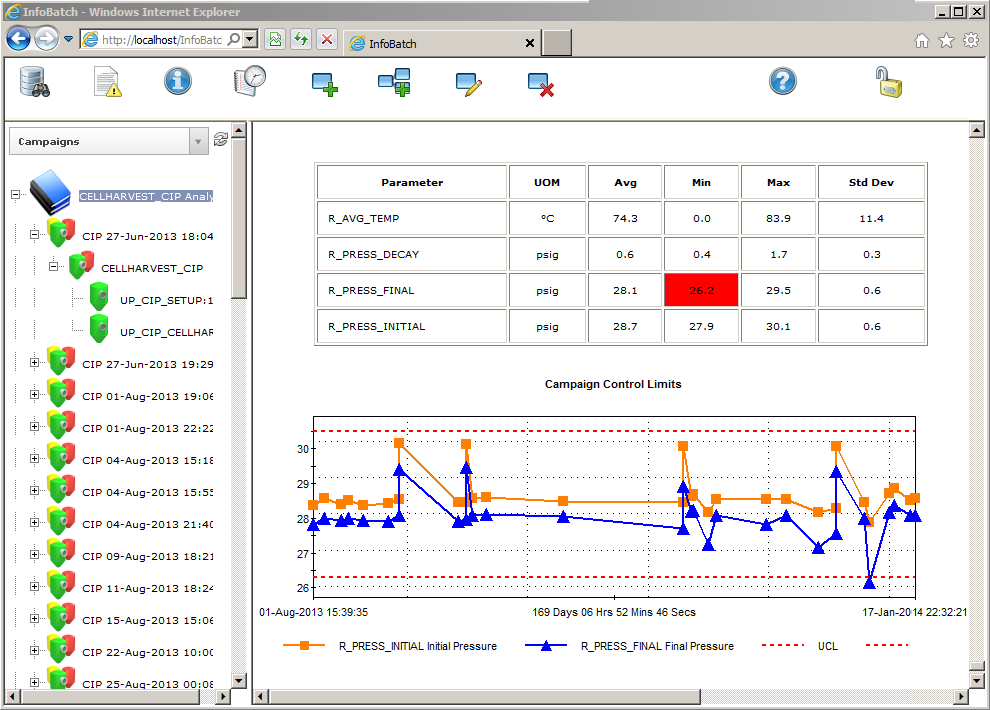
Process Analytical Technology (PAT) is becoming increasingly important in pharmaceuticals manufacturing. As regulatory authorities continue to push for greater certainty in process outcomes and higher levels of product quality, the demand for PAT looks set for further growth.
The ultimate goal of a PAT solution is to provide a deep process understanding such that variability is understood and controlled, and quality attributes can be accurately predicted. Using InfoBatch, manufacturers can document and analyze each step within a process. Process parameters that are out of defined limits can be identified immediately, providing the ability to easily correct, understand and improve overall production. Data is collected in a user-friendly format, providing manufacturers with the ability to make informed decisions in improving the process of future batches.
Request Demo